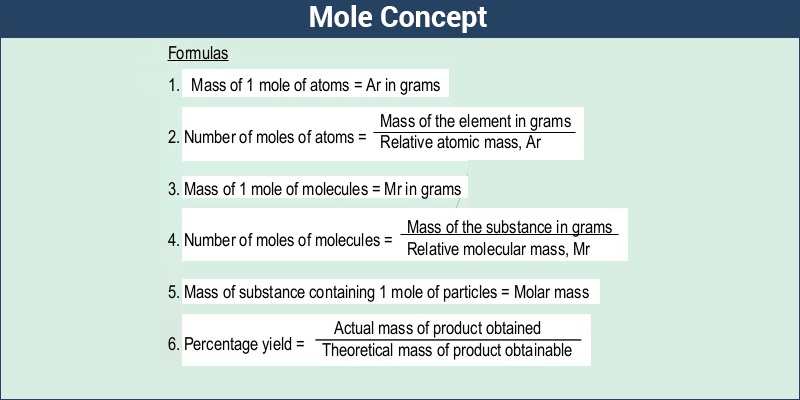
MOLAR MASS Molar mass of a substance = mass in grams of one mole of the substance. A compound's molar mass is NUMERICALLY equal to its formula mass. Formula. - ppt download

Relating Mass to Numbers of Atoms The MOLE. Is the SI unit for amount of a substance Abbreviated as mol Amount of a substance that contains as many atoms. - ppt download