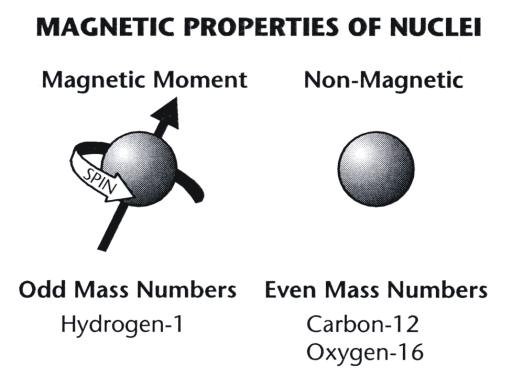
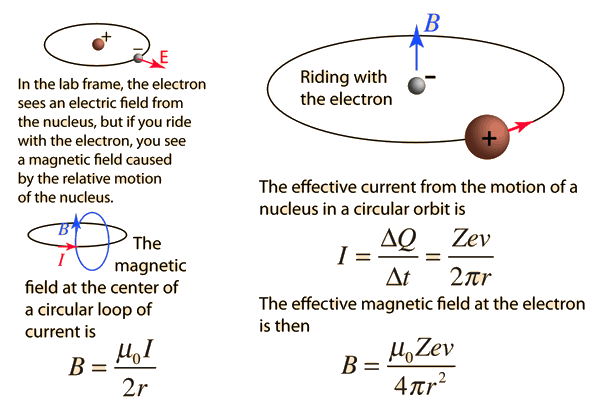
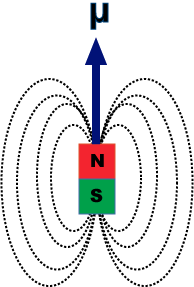
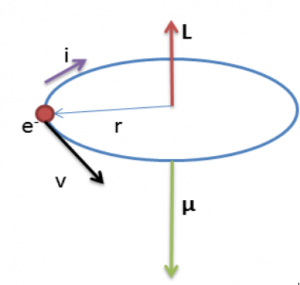
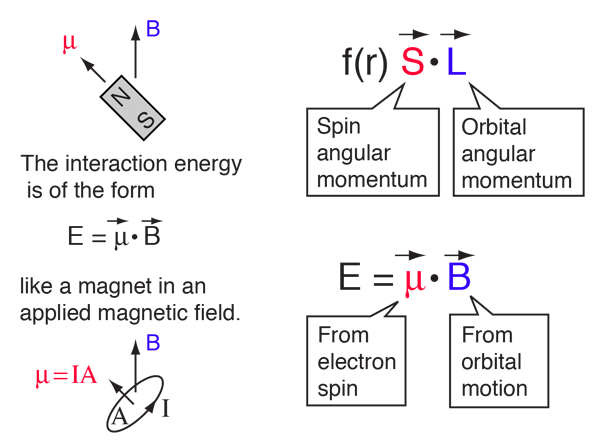
An electron revolving in an orbit of radius 0.5 A in a hydrogen atom executes 10^16 revolutions per second. The magnetic moment of electron due to its orbital motion will be

In a hydrogen atom, the binding energy of the electron in the ground state is E1. Then the frequency of revolution of nth electron in the nth orbits is

In a hydrogen atom, an electron of mass m and charge e revolves in an orbit of radius r making n revolutions per second. If the mass of hydrogen nucleus is M,

quantum mechanics - Ignoring spin, what is its orbital magnetic moment of an electron in a hydrogen atom in the 2p orbital? - Physics Stack Exchange
The minimum magnetic dipole moment of electron in hydrogen atom is - Sarthaks eConnect | Largest Online Education Community
Magnetic Moment Physics Of Magnetic Resonance Imaging Magnetic Field Hydrogen Atom, PNG, 1233x874px, Magnetic Moment, Area,
Calculate the magnetic dipole moment corresponding to the motion of the electron in the ground state of a hydrogen atom. - Sarthaks eConnect | Largest Online Education Community

SOLVED: A hydrogen atom in the 5 g state is placed in a magnetic field of 0.600 T that is in the z -direction. (a) Into how many levels is this state