Novartis' Cosentyx Moves Closer to a 4th Indication with Positive Data in Spondyloarthritis Trial – Intelligence Pharma
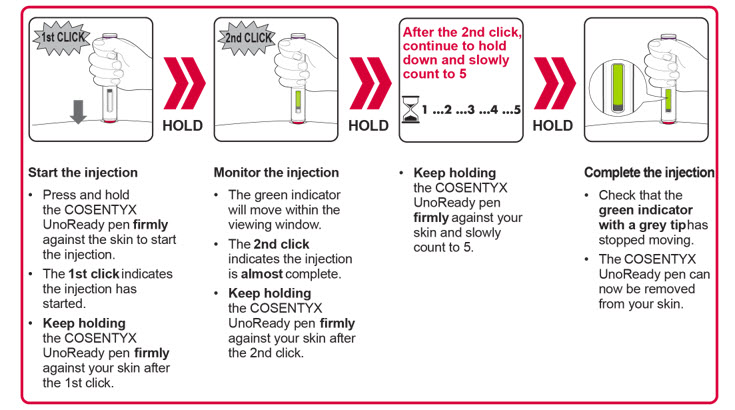
INSTRUCTIONS FOR USE COSENTYX® [koe-sen-tix] (secukinumab) injection, for subcutaneous use 300 mg/2 mL single-dose prefilled sy
INSTRUCTIONS FOR USE COSENTYX® [koe-sen-tix] (secukinumab) injection, for subcutaneous use 300 mg/2 mL single-dose prefilled sy